Jenlab supplies advanced endotoxin testing systems and reagents from Charles River Laboratories. Our products support pharmaceuticals, radio-pharmaceuticals, biotechnology and research laboratories with rapid and reliable endotoxin detection solutions.
The Endosafe® nexgen-PTS is a portable endotoxin testing system designed for rapid, point-of-use analysis. This handheld spectrophotometer uses disposable cartridges to deliver accurate quantitative endotoxin results in approximately 15 minutes, making it ideal for laboratory and manufacturing environments.
• Portable handheld endotoxin testing system
• Rapid quantitative results in ~15 minutes
• Disposable cartridges.
• 21 CFR Part 11 Compliant.


The Endosafe® nexgen-MCS is a benchtop endotoxin testing system designed for laboratories that require higher sample throughput. The system allows multiple samples to be tested simultaneously using Endosafe cartridge technology, providing rapid quantitative endotoxin results in approximately 15 minutes.
• Multi-sample endotoxin testing system
• Tests up to 5 samples simultaneously
• Rapid quantitative results in ~15 minutes
• 21 CFR Part 11 Compliant.
• Used with Endoscan V software.
The Endosafe® PRS 3 Microplate Reader is a spectrophotometer designed for endotoxin testing using a 96-well microplate format. It supports traditional kinetic endotoxin assays and integrates with EndoScan-V™ software for efficient analysis and reporting.
• 96-well microplate endotoxin testing system
• Compatible with Endosafe® LAL reagents
• Integration with EndoScan-V™ software
• Designed for laboratory endotoxin testing workflows


Endosafe® LAL Cartridges provide rapid endotoxin testing using a kinetic chromogenic LAL method. Each disposable cartridge contains the reagents and controls required for testing, allowing laboratories to obtain fast and reliable results with minimal preparation.
• Cartridge-based kinetic chromogenic endotoxin testing & Inhibition and Enhancement.
• Pre-loaded with LAL reagents and controls
• Rapid quantitative results in ~15 minutes
• Compatible with Endosafe testing systems
Endosafe® Trillium™ rCR is an animal-free endotoxin testing reagent that uses recombinant technology to simulate the traditional LAL reaction. It provides rapid and reliable endotoxin detection and is available in both Endosafe cartridge format and lysate (vial) format for traditional kinetic testing methods.
• Animal-free recombinant endotoxin testing reagent
• Available in cartridge and lysate (vial) formats
• Rapid and reliable endotoxin detection
• Compatible with Endosafe systems and traditional assays


Limulus Amebocyte Lysate (LAL) reagents are used to detect and measure bacterial endotoxin contamination in pharmaceutical products, biological materials, and medical devices. LAL testing is one of the most sensitive and widely used methods for bacterial endotoxin testing in quality control laboratories.
• Highly sensitive detection of bacterial endotoxins
• Suitable for gel-clot, kinetic, and chromogenic assay methods
• Includes reagents, buffers, standards, and testing accessories
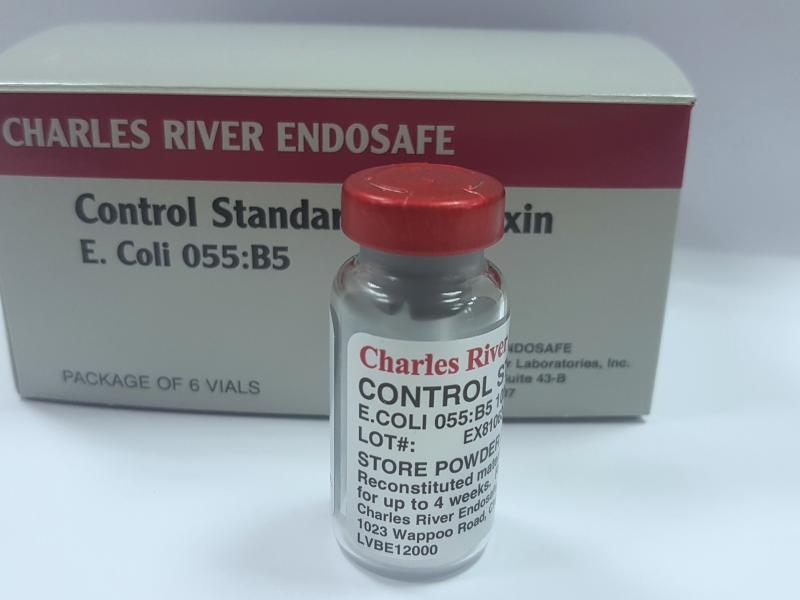
Control Standard Endotoxin (CSE) is a purified endotoxin standard used to verify and calibrate endotoxin testing systems. It is standardised against reference endotoxin materials to ensure accurate and reliable test results during bacterial endotoxin testing.
• Used to validate endotoxin testing assays
• Standardised against reference endotoxin standards
• Ensures accurate and reliable test performance
• Suitable for LAL-based endotoxin testing methods